Original Source(s)
Related Content
Retinal Ischemia as Stroke Warning
Retrospective study investigates links between acute ischemic ocular events and increased stroke risk
May 8, 2026
-
3 min

Vamikibart Shows Early Safety in Uveitic Macular Edema
Phase 1 nonrandomized trial reported no drug-related serious adverse events, but efficacy remains unproven
by Kerri Miller
May 7, 2026
-
3 min
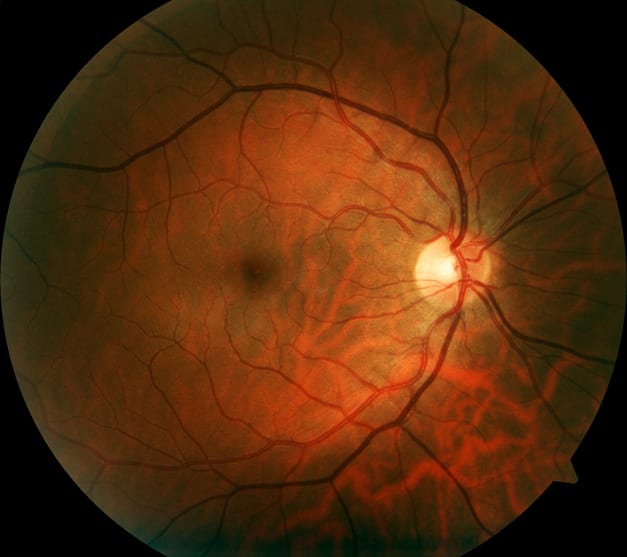
Chatbots Enter Retinal Care
New research shows how conversational AI might improve patient understanding and engagement in retinal detachment pathways
May 7, 2026
-
3 min


