New Label Expansion in Generalized Myasthenia Gravis
-
By
-
Kathryn Wighton
-
May 11, 2026
-
2 min
By
Kathryn Wighton
May 11, 2026
Diagnosing Parkinson’s disease has long depended primarily on clinical expertise — careful neurologic examination, longitudinal symptom assessment and the nuanced interpretation of movement abnormalities.
Epilepsy remains a life-altering condition, particularly due to the unpredictable nature of seizures and their cumulative impact on cognition, independence and quality of life.
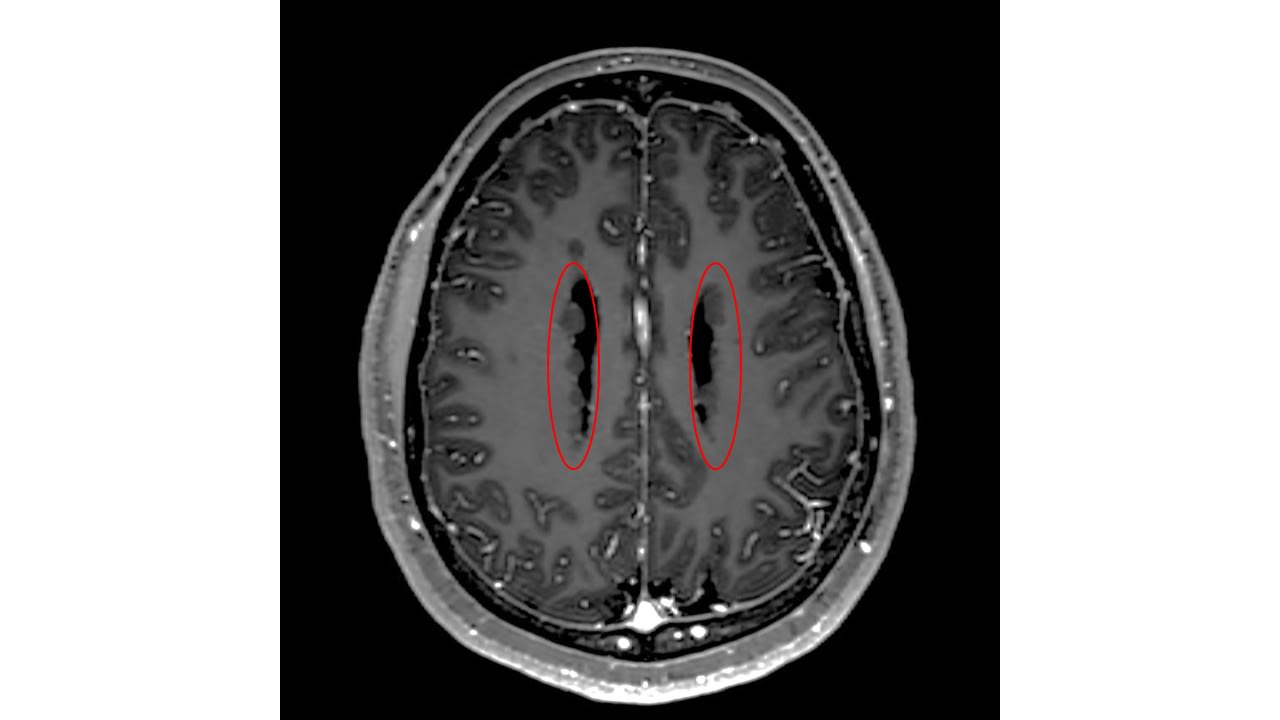
Integrated neuromonitoring approaches may help identify neurologic complications earlier in patients admitted to ICU.
May 7, 2026
